The project
ENDOSCAPE is a collaborative biotechnology project aimed at developing a beyond state of the art clinically applicable gene delivery technology. The ENDOSCAPE technology will have a major impact on the therapeutic opportunities for current and future drugs for a broad range of diseases and large patient groups.
The non-viral based ENDOSCAPE technology will enhance therapeutic efficacy in a more cost-effective manner thereby reducing costs of healthcare, improving the health of patients worldwide, and strengthening the competitive landscape of the EU in the worldwide quest for such an advanced technology.
The project is funded by the European Commission with about 7 million € under the Horizon 2020 initiative, with the grant agreement ID: 825730. Project results can be found here: ENDOSCAPE @ EC Cordis
For more details visit the ENDOSCAPE vision.
Follow us at LinkedIn: https://www.linkedin.com/company/endoscapeproject/
Vision
ENDOSCAPE will overcome a longstanding and major bottleneck in the field of gene delivery, namely efficient, safe and cost-effective transfer of gene therapeutic products into the cytosol.
The ENDOSCAPE technology will be designed to allow targeting of any addressable cell type with all known genetic agents, thereby ensuring better patient therapy not limited to inherited disorders, but also for cancer therapy and therefore of importance for large patient groups.
Consortium
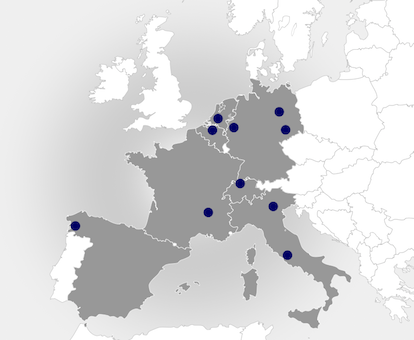
The ENDOSCAPE consortium brings together an interdisciplinary chain of internationally recognized European partners with academic and industrial technologies and expertise for the development of the ENDOSCAPE gene delivery technology, as well as warranting future exploitation and commercialization.
You can find out more about the individual partners of the ENDOSCAPE consortium and their contact information.
